China Standardizes Medical Consumable Fees for Refined Management
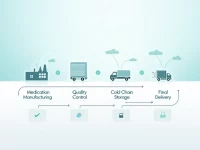
By 2026, medical consumables management enters an era of precision. Several regions, including Henan, have released chargeable medical consumables catalogs and updated medical insurance catalogs, linking them to centralized procurement results, project guidelines, and payment policies. Medical insurance payment is becoming more refined, with local governments setting price limits and differentiated co-payment ratios. At the national level, unified management is being promoted. Enterprises need to adapt to policy changes and adjust their product strategies.